- HOME
- VENUE
- RSVP
- REGISTRY
- CONTACT
- Loaris trojan remover v2-0-12
- Convergence plot nonmem pirana
- Wmma 5 wine
- Kirby return to dreamland iso ntsc mega
- What is the best flight simulator
- Extreme car driving simulator mod apk hack download
- Ooouuu william singe lyrics
- Audirvana plus
- Cookie run ovenbreak 2 year anniversary event
- Mach3 cnc and sheetcam for sale
- Magix music maker downloaden
- What happened to the lone ranger horse silver

The LTG population pharmacokinetic model developed in this study may be a reliable method for individualising the LTG dosing regimen in paediatric and young adult patients on combination therapy during therapeutic drug monitoring. VPA was found to decrease the LTG CL/F by 87.6 % compared to co-therapy with only CBZ. If a patient was concomitantly treated with CBZ + VPA, the CL/F decreased on average by 69.5 % relative to LTG + CBZ co-therapy. The final LTG population model included the effects of concomitant drugs and patient’s weight (WT) which stratified the population into three groups: ≤25 kg, >25 to 25 to <60 kg or ≥60 kg who was concomitantly treated with CBZ was estimated to be 3.28, 4.23, or 7.15 l/h, respectively.
CONVERGENCE PLOT NONMEM PIRANA SOFTWARE
Data were modelled, and the final model was evaluated using NONMEM and auxiliary software tools. A total of 70 blood samples corresponding to trough levels were available for analysis. The study included 53 patients (age range 3–35 years) who were concomitantly treated with carbamazepine (CBZ) and/or valproic acid (VPA). doi: 10.1007/s1092-y.The purpose of the study was to examine and describe adjunctive lamotrigine (LTG) pharmacokinetics in paediatric and young adult patients using a nonlinear mixed effects modelling (NONMEM) approach. Comparing the performance of FOCE and different expectation-maximization methods in handling complex population physiologically-based pharmacokinetic models. Comparison of Nonmem 7.2 estimation methods and parallel processing efficiency on a target-mediated drug disposition model. Derivation of various NONMEM estimation methods. Basic concepts in population modeling, simulation, and model-based drug development-part 2: introduction to pharmacokinetic modeling methods. Interpreting population pharmacokinetic-pharmacodynamic analyses - a clinical viewpoint. EM-based estimation methods can be considered for adapting to the specific needs of a modeling project at later steps of modeling.Įstimation methods Few subjects First-order conditional estimation with interaction Markov chain Monte Carlo Bayesian NONMEM.ĭuffull SB, Wright DF, Winter HR. The classical FOCE-I method appeared to estimate the PK parameters more reliably than the BAYES method when using a simple model and data containing only a few subjects. Similar performance of the estimation methods was observed with theophylline dataset.

In general, estimates of random-effect parameters showed significant bias and imprecision, irrespective of the estimation method used and the level of IIV. The rRMSE and REE values of all parameter (fixed effect and random effect) estimates showed that all four methods performed equally at the lower IIV levels, while the FOCE-I method performed better than other EM-based methods at higher IIV levels (greater than 30%). NONMEM software assisted by Pirana, PsN, and Xpose was used to estimate population PK parameters, and R program was used to analyze and plot the results. A case study was performed with a clinical data of theophylline available in NONMEM distribution media.

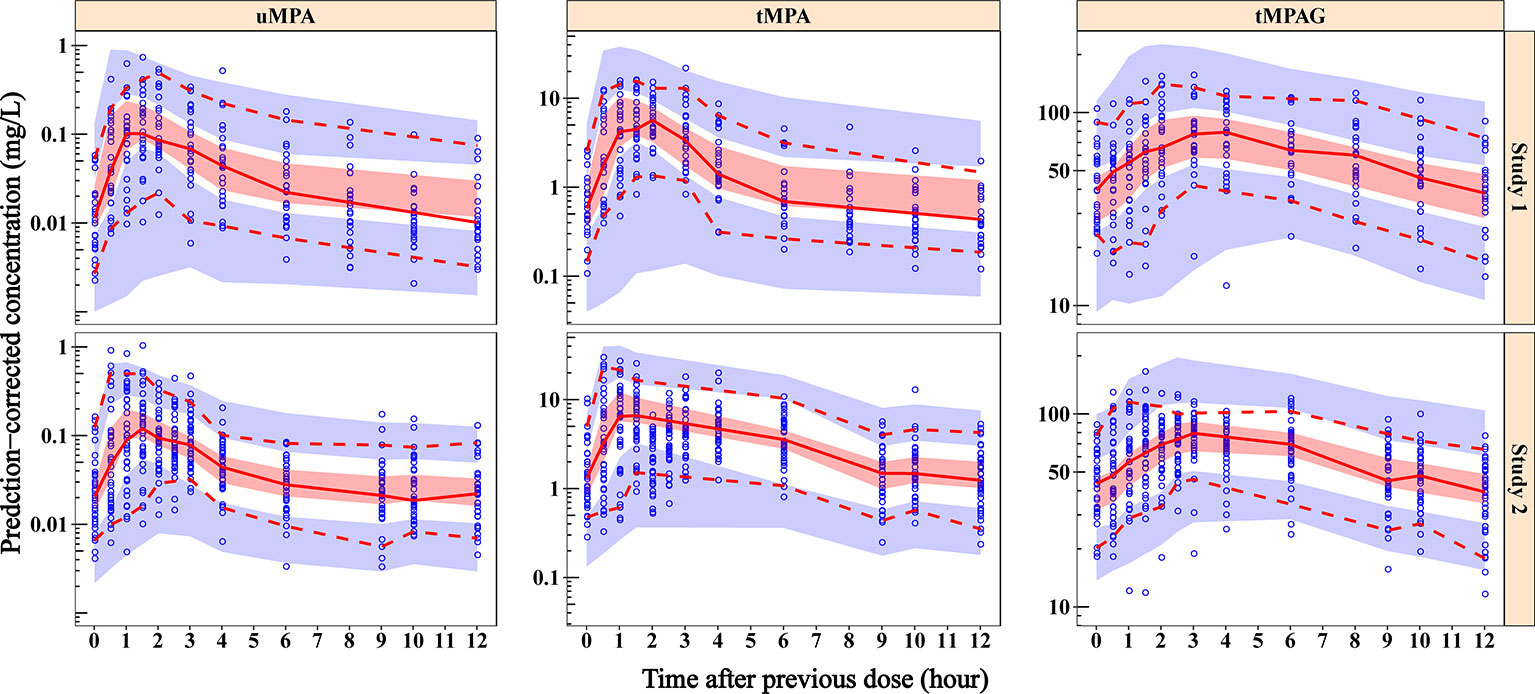
Relative root mean squared error (rRMSE) and relative estimation error (REE) were used to analyze the differences between true and estimated values. A stochastic simulation and estimation (SSE) study was performed to simultaneously simulate data sets and estimate the parameters using four different methods: FOCE-I only, BAYES(C) (FOCE-I and BAYES composite method), BAYES(F) (BAYES with all true initial parameters and fixed ω 2 ), and BAYES only. In this study, 100 data sets were simulated with eight sampling points for each subject and with six different levels of IIV (5%, 10%, 20%, 30%, 50%, and 80%) in their PK parameter distribution. In this study, the performance of a classical first-order conditional estimation with interaction (FOCE-I) and expectation maximization (EM)-based Markov chain Monte Carlo Bayesian (BAYES) estimation methods were compared for estimating the population parameters and its distribution from data sets having a low number of subjects. Exploratory preclinical, as well as clinical trials, may involve a small number of patients, making it difficult to calculate and analyze the pharmacokinetic (PK) parameters, especially if the PK parameters show very high inter-individual variability (IIV).